Plasma Technology
– What is Plasma?
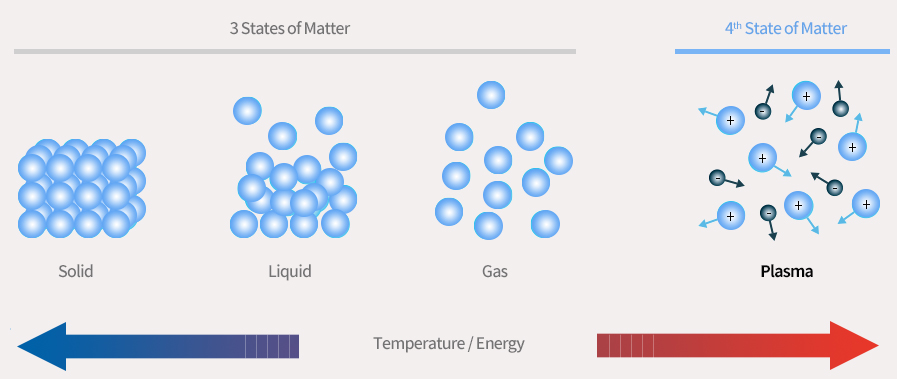
Plasma refers to ‘ionized gas.’ When high energy is applied to the gas atoms or molecules, the electrons get knocked off the particles, ionizing the gas. As a result, the particles are divided into positive and negative ions, radicals, and photons – forming plasma.
The plasma shows apparent neutrality due to the same number of positively and negatively charged particles (Quasi-Neutral). Since plasma possesses free moving electrons and charged particles, it is highly responsive to electric and magnetic fields (Collective Behavior).
Nowadays, plasma plays an essential role in many industrial fields. In semi-conductor manufacturing, more than 70% of the process uses plasma. By using the chemical and physical properties of plasma, we can change the surface properties of materials. It is a future-oriented technology with new applications continually being developed.
The Fourth State of Matter
Plasma systems divide into vacuum and atmospheric types.

Femtoscience Inc. has a unique chamber technology and design to provide effective and reliable systems for researches and businesses.

Plasma is generated in the 100 to 760 Torr (Atmospheric) pressure range. The atmospheric system is known for its high throughput compared to vacuum systems.